 
Although expected to be weaker than the standard London forces, these interactions display the same R$^6$ distance scaling. Within a second-order perturbative approach, these contributions manifest as an effective intermolecular exchange interaction. The model assumes that the same fluctuating electric fields responsible for vdW forces can induce a magnetic response via a Rashba-like term, so that an spin-orbit field acting on molecule B is generated by the electric field arising from charge density fluctuations in molecule A (and viceversa). Motivated by it, we propose in this investigation to describe the mutual induction of charge and spin-density fluctuations in a pair A-B of chiral molecules by a simple physical model. The discovery of the Chirality-Induced Spin Selectivity (CISS) effect in recent years has led to an additional twist in the study of chiral molecular systems, showing a close relation between spin and molecular geometry. Order the intermolecular forces (dipole-dipole, London Dispersion, Ionic, and hydrogen bonding from weakest to strongest) a) Dipole-dipole, London Dispersion, ionic, and hydrogen bonding b) London dispersion, dipole-dipole, hydrogen-bonding, and ionic c) Hydrogen bonding, dipole-dipole, London dispersion and ionic d) Dipole-dipole, ionic, London Dispersion, and hydrogen-bonding e) London. 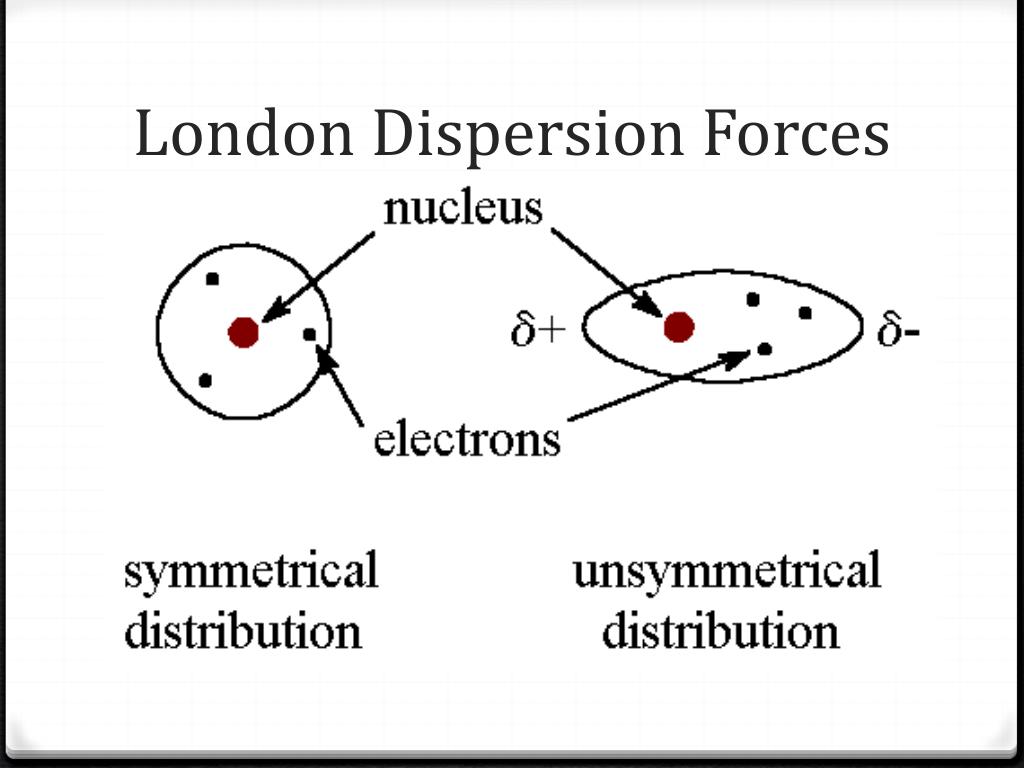
Additional applications include the interparticle forces that can be measured by direct tech-niques, such as atomic force microscopy. However, it was only in the 1960s that it was recognized that for molecules lacking an inversion center such as chiral and helical molecules, there are chirality-sensitive corrections to the dispersion forces proportional to the rotatory power known from the theory of circular dichroism and with the same distance scaling law R-6 as the London energy. London dispersion forces makes them a factor in a wide spectrum of problems they have been in evidence since the pioneering work of Young and Laplace on wetting, contact angles, and surface energies. The theory of dispersion forces was developed by London in the early years of quantum mechanics. Geyer and 5 other authors Download PDF Abstract:Dispersion interactions are one of the components of van der Waals (vdW) forces, which play a key role in the understanding of intermolecular interactions in many physical, chemical and biological processes. Download a PDF of the paper titled The contribution of intermolecular spin interactions to the London dispersion forces between chiral molecules, by M. Thus London dispersion forces are responsible for the general trend toward higher boiling points with increased molecular mass and greater surface area in a homologous series of compounds, such as the alkanes (part (a) in Figure 11.6 'Mass and Surface Area Affect the Strength of London Dispersion Forces'). These London dispersion forces are often found in the halogens (e.g., F2 and I2), the noble gases (e.g., Ne and Ar), and in other non-polar molecules, such as. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
